By Meghan McCarthy
The morning of March 21, 2019, began unremarkably for Nancy and Mike Childs.
After waking up, they began their simple morning routine. Then they turned on the news, and their world shifted.
Reports informed them that the Phase III trial of the drug aducanumab run by Biogen and Eisai — a trial in which Mike was participating — had ceased, effective immediately. It was one thing to have their hopes of a successful treatment dashed without warning; it was another to have learned this news from the television.
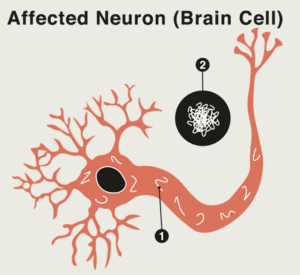
After Mike Childs was diagnosed with mild cognitive impairment (MCI) in 2014, he decided to enroll in the aducanumab trial, which aimed at slowing the progression of symptoms from MCI or Alzheimer’s disease (AD). Mike enrolled with hope for himself and the future of his disease.
The trial became more than a potential treatment; it became a community for the Childses.